An Update on the Genetics of Polycystic Ovary Syndrome
PCOS and Genetics
DOI:
https://doi.org/10.18311/jer/2023/34654Keywords:
Etiopathogenesis, Genetic Variants, GWAS, Next-Generation Sequencing, Polycystic Ovary Syndrome, Whole Exome SequencingAbstract
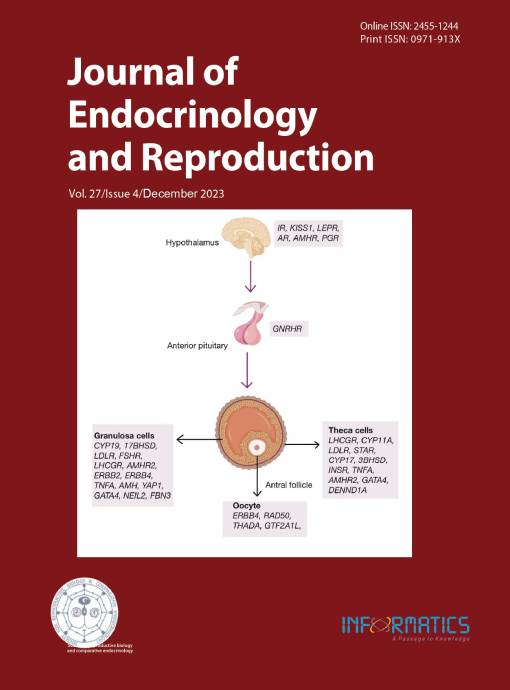
PCOS is a common endocrinopathy among women of reproductive age, with a worldwide prevalence of 8 to 13%, depending on the criteria used for diagnosis. It is characterized by a constellation of features, including oligo/anovulation, clinical and/or biochemical hyperandrogenism, and polycystic ovarian morphology. PCOS is one of the common causes of female infertility. It is also associated with metabolic derangements, including obesity, insulin resistance, and compensatory hyperinsulinemia, which increase the likelihood of developing type 2 diabetes mellitus. Despite extensive research, the etiology of PCOS remains largely unknown. It seems likely that the hypothalamic-pituitary-ovarian axis dysfunction, partial folliculogenesis arrest, insulin resistance, and ovarian and adrenal androgen secretion may play a role in the pathogenesis of PCOS. Familial clustering of the cases of PCOS points to a genetic component linked with it. The initial genetic studies suggest an autosomal dominant pattern of inheritance of the disorder in some families; however, most studies support multifactorial origin. Since PCOS is a complex trait, the typical form of inheritance of PCOS follows a non-Mendelian pattern and involves complex genetic mechanisms. Studies involving linkage and association have suggested a connection between genetic variations and the risk of developing PCOS in certain families or populations. Through genome-wide association studies and next-generation sequencing techniques, several candidate genes have been identified that play a role in the etiopathogenesis of the disorder. Pathogenic variants of various genes such as INSR, IRS1, GHRL, LDLR, MC4R, ADIPOQ, UCP1, UCP2, UCP3, FTO, PCSK9, FBN3, NEIL2, FDFT1, PCSK9, CYP11, CYP17, CYP21, HSD17, STAR, POR, AKR1C3, AMH, AMHR2, INHBA, AR, SHBG, LHR, FSHR, FSH β, SRD5A, GATA4, THADA, YAP1, ERBB2, DENND1A, FEM1B, FDFT1, NEIL2, TCF7L2, etc. in some PCOS cases are linked as underlying etiologic associations. This review aims to provide insight into the current genetic knowledge about PCOS. Discovering the genetic factors and pathways involved in the disorder will help us better comprehend the underlying mechanisms of the disorder.
Downloads
Metrics
Downloads
Published
How to Cite
Issue
Section
References
Azziz R, Carmina E, Dewailly D, et al. The Androgen Excess and PCOS Society criteria for the polycystic ovary syndrome: the complete task force report. Fertil Steril. 2009; 91(2):456- 88. https://doi.org/10.1016/j.fertnstert.2008.06.035
Teede H, Deeks A, Moran L. Polycystic ovary syndrome: a complex condition with psychological, reproductive and metabolic manifestations that impacts on health across the lifespan. BMC Med. 2010; 8:41. https://doi. org/10.1186/1741-7015-8-41
Azziz R, Carmina E, Chen Z, et al. Polycystic ovary syndrome. Nat Rev Dis Primers. 2016; 2:16057. https://doi. org/10.1038/nrdp.2016.57
Teede HJ, Misso ML, Deeks AA, et al. Assessment and management of polycystic ovary syndrome: Summary of an evidence-based guideline. Med J Aust. 2011; 195:S65–112. https://doi.org/10.5694/mja11.10915
Cooney LG, Lee I, Sammel MD, Dokras A. High prevalence of moderate and severe depressive and anxiety symptoms in polycystic ovary syndrome: A systematic review and meta-analysis. Hum Reprod. 2017; 32:1075–91. https://doi. org/10.1093/humrep/dex044
Teede HJ, Misso ML, Costello MF, et al. International PCOS Network. Recommendations from the international evidencebased guideline for the assessment and management of polycystic ovary syndrome. Hum Reprod. 2018; 33(9):1602- 18. https://doi.org/10.1093/humrep/dey256
Teede HJ, Misso ML, Costello MF, et al. International PCOS Network. Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Clin Endocrinol (Oxf). 2018; 89(3):251-68. https://doi.org/10.1111/cen.13795
Chen ZJ, Zhao H, He L, Shi Y, Qin Y, Shi Y, Li Z, You L, Zhao J, Liu J, et al. Genome-wide association study identifies susceptibility loci for polycystic ovary syndrome on chromosome 2p16.3, 2p21 and 9q33.3. Nat Genet. 2011; 43:55–9. https://doi.org/10.1038/ng.732
Gorsic LK, Kosova G, Werstein B, et al. Pathogenic antiMüllerian hormone variants in polycystic ovary syndrome. J Clin Endocrinol Metab. 2017; 102(8):2862-72. https://doi.org/10.1210/jc.2017-00612
Shi Y, Zhao H, Shi Y, Cao Y, Yang D, Li Z, et al. Genomewide associationstudy identifies eight new risk loci for polycystic ovary syndrome. Nat Genet. 2012; 44(9):1020. https://doi.org/10.1038/ng.2384
Hayes MG, Urbanek M, Ehrmann DA, et al. Genomewide association of polycystic ovary syndrome implicates alterations in gonadotropin secretion in European ancestry populations. Nat Commun. 2015; 6(1):7502. https://doi. org/10.1038/ncomms8502
Day F, Karaderi T, Jones MR, et al. Large-scale genomewide meta-analysis of polycystic ovary syndrome suggests shared genetic architecture for different diagnosis criteria. PLoS Genet. 2018; 14(12):e1007813.
Zhang Y, Ho K, Keaton JM, et al. A genome-wide association study of polycystic ovary syndrome identified from electronic health records. Am J Obstet Gynecol. 2020; 223(4):559.e1-55. e21. https://doi.org/10.1016/j.ajog.2020.04.004
Vink JM, Sadrzadeh S, Lambalk CB, Boomsma DI. Heritability of polycystic ovary syndrome in a Dutch twinfamily study. J Clin Endocrinol Metab. 2006; 91(6):2100-4. https://doi.org/10.1210/jc.2005-1494
Dunaif A. Perspectives in polycystic ovary syndrome: from hair to eternity. J Clin Endocrinol Metab. 2016; 101(3):759- 68. https://doi.org/10.1210/jc.2015-3780
Manolio TA, Collins FS, Cox NJ, et al. Finding the missing heritability of complex diseases. Nature. 2009; 461(7265):747-53. https://doi.org/10.1038/nature08494
Cirulli ET, Goldstein DB. Uncovering the roles of rare variants in common disease through whole-genome sequencing. Nat Rev Genet. 2010; 11(6):415-25. https://doi. org/10.1038/nrg2779
Cooper HE, Spellacy WN, Prem KA, Cohen WD. Hereditary factors in the Stein-Leventhal syndrome. Am J Obstet Gynecol. 1968; 100(3):371-87. https://doi. org/10.1016/S0002-9378(15)33704-2
Judd HL, Scully RE, Herbst AL, Yen SS, Ingersol FM, Kliman B. Familial hyperthecosis: comparison of endocrinologic and histologic findings with polycystic ovarian disease. Am J Obstet Gynecol. 1973; 117(7):976-82. https://doi. org/10.1016/0002-9378(73)90071-9
Cohen PN, Givens JR, Wiser WL, et al. Polycystic ovarian disease, maturation arrest of spermiogenesis, and Klinefelter’s syndrome in siblings of a family with familial hirsutism. Fertil Steril. 1975; 26(12):1228-38. https://doi. org/10.1016/S0015-0282(16)41539-6
Ferriman D, Purdie AW. The inheritance of polycystic ovarian disease and a possible relationship to premature balding. Clin Endocrinol (Oxf). 1979; 11(3):291-300. https://doi.org/10.1111/j.1365-2265.1979.tb03077.x
Givens JR. Familial polycystic ovarian disease. Endocrinol Metab Clin North Am. 1988; 17(4):771-83. https://doi. org/10.1016/S0889-8529(18)30409-2
Legro RS, Driscoll D, Strauss JF 3rd, Fox J, Dunaif A. Evidence for a genetic basis for hyperandrogenemia in polycystic ovary syndrome. Proc Natl Acad Sci USA. 1998; 95(25):14956-60. https://doi.org/10.1073/pnas.95.25.14956
Kashar-Miller M, Azziz R. Heritability and the risk of developing androgen excess. J Steroid Biochem Mol Biol. 1999; 69(1-6):261-8. https://doi.org/10.1016/S0960- 0760(99)00043-6
Govind A, Obhrai MS, Clayton RN. Polycystic ovaries are inherited as an autosomal dominant trait: analysis of 29 polycysticovary syndrome and 10 control families. J Clin Endocrinol Metab. 1999; 84(1):38-43. https://doi. org/10.1210/jcem.84.1.5382
Franks S, Webber LJ, Goh M, et al. Ovarian morphology is a marker of heritable biochemical traits in sisters with polycystic ovaries. J Clin Endocrinol Metab. 2008; 93(9):3396-402. https://doi.org/10.1210/jc.2008-0369
Lunde O, Magnus P, Sandvik L, Høglo S. Familial clustering in the polycystic ovarian syndrome. Gynecol Obstet Invest. 1989; 28(1):23-30. https://doi.org/10.1159/000293493
Carey AH, Chan KL, Short F, White D, Williamson R, Franks S. Evidence for a single gene effect causing polycystic ovaries and male pattern baldness. Clin Endocrinol (Oxf). 1993; 38(6):653-8. https://doi.org/10.1111/j.1365-2265.1993. tb02150.x
Kahn CR, Flier JS, Bar RS, et al. The syndromes of insulin resistance and acanthosis nigricans. Insulin-receptor disorders in man. N Engl J Med. 1976; 294(14):739-45. https://doi.org/10.1056/NEJM197604012941401
Taylor SI, Cama A, Accili D, et al. Mutations in the insulin receptor gene. Endocr Rev. 1992; 13(3):566-95. https://doi. org/10.1210/edrv-13-3-566
Legro RS, Bentley-Lewis R, Driscoll D, Wang SC, Dunaif A. Insulin resistance in the sisters of women with polycystic ovary syndrome: association with hyperandrogenemia rather than menstrual irregularity. J Clin Endocrinol Metab. 2002; 87(5):2128-33. https://doi.org/10.1210/jcem.87.5.8513
Sorbara LR, Tang Z, Cama A, et al. Absence of insulin receptor gene mutations in three insulin-resistant women with the polycystic ovary syndrome. Metabolism. 1994; 43(12):1568- 74. https://doi.org/10.1016/0026-0495(94)90018-3
Stuart CA, Peters EJ, Prince MJ, Richards G, Cavallo A, Meyer WJ 3rd. Insulin resistance with acanthosis nigricans: the roles of obesity and androgen excess. Metabolism. 1986; 35(3):197- 205. https://doi.org/10.1016/0026-0495(86)90201-5
Li J, et al. Transmission of polycystic ovary syndrome susceptibility single-nucleotide polymorphisms and their association with phenotype changes in offspring. Hum. Reprod. 2020; 35:1711–8. https://doi.org/10.1093/humrep/ deaa125
Jahanfar S, Eden JA, Warren P, Seppälä M, Nguyen TV. A twin study of polycystic ovary syndrome. Fertil Steril. 1995; 63(3):478-86. https://doi.org/10.1016/S0015-0282(16)57412-3
Franks S, Gharani N, Waterworth D, Batty S, White D, Williamson R, McCarthy M. The genetic basis of polycystic ovary syndrome. Hum Reprod. 1997; 12(12):2641-8. https://doi.org/10.1093/humrep/12.12.2641
Visscher PM, Hill WG, Wray NR. Heritability in the genomics era–concepts and misconceptions. Nat Rev Genet. 2008; 9(4):255-66. https://doi.org/10.1038/nrg2322
Conley D, Rauscher E, Dawes C, Magnusson PK, Siegal ML. Heritability and the equal environments assumption: evidence from multiple samples of misclassified twins. Behav Genet. 2013; 43(5):415-26. https://doi.org/10.1007/ s10519-013-9602-1
Harris JA, Vernon PA, Boomsma DI. The heritability of testosterone: a study of Dutch adolescent twins and their parents. Behav Genet. 1998; 28(3):165-71. https://doi. org/10.1023/A:1021466929053
Hoekstra RA, Bartels M, Boomsma DI. Heritability of testosterone levels in 12-year-old twins and its relation to pubertal development. Twin Res Hum Genet. 2006; 9(4): 558-65. https://doi.org/10.1375/twin.9.4.558
Coviello AD, Zhuang WV, Lunetta KL, et al. Circulating testosterone and SHBG concentrations are heritable in women: the Framingham Heart Study. J Clin Endocrinol Metab. 2011; 96(9):E1491-5. https://doi.org/10.1210/jc.2011- 0050
Yildiz BO, Goodarzi MO, Guo X, Rotter JI, Azziz R. Heritability of dehydroepiandrosterone sulfate in women with polycystic ovary syndrome and their sisters. Fertil Steril. 2006; 86(6):1688-93. https://doi.org/10.1016/j.fertnstert.2006.05.045
Stein IF, Leventhal ML. Amenorrhea associated with bilateral polycystic ovaries. Am J Obstet Gynecol. 1935; 29:181-91. https://doi.org/10.1016/S0002-9378(15)30642-6
Dumesic DA, Abbott DH, Sanchita S, Chazenbalk GD. Endocrine-metabolic dysfunction in polycystic ovary syndrome: an evolutionary perspective. Curr Opin Endocr Metab Res. 2020; 12:41-8. https://doi.org/10.1016/j. coemr.2020.02.013
Marshall JC, Eagleson CA. Neuroendocrine aspects of polycystic ovary syndrome. Endocrinol Metab Clin North Am. 1999; 28(2):295-324. https://doi.org/10.1016/S0889- 8529(05)70071-2
Banaszewska B, Spaczynski RZ, Pelesz M, Pawelczyk L. Incidence of elevated LH/FSH ratio in polycystic ovary syndrome women with normo and hyperinsulinemia. Rocz Akad Med Bialymst. 2003; 48:131–4.
Dale PO, Tanbo T, Vaaler S, Abyholm T. Body weight, hyperinsulinemia, and gonadotropin levels in the polycystic ovarian syndrome: evidence of two distinct populations. Fertil Steril. 1992; 58:487–91. https://doi.org/10.1016/ S0015-0282(16)55249-2
Burt Solorzano CM, McCartney CR, Blank SK, Knudsen KL, Marshall JC. Hyperandrogenaemia in adolescent girls: origins of abnormal gonadotropin releasing hormone secretion. BJOG. 2010; 117:143–9. https://doi.org/10.1111/ j.1471-0528.2009.02383.x
Eagleson CA, Gingrich MB, Pastor CL, Arora TK, Burt CM, Evans WS, Marshall JC. Polycystic ovarian syndrome: evidence that flutamide restores sensitivity of the gonadotropinreleasing hormone pulse generator to inhibition by estradiol and progesterone. J Clin Endocrinol Metab. 2000; 85(11):4047- 52. https://doi.org/10.1210/jc.85.11.4047
Ehrmann DA, Barnes RB, Rosenfield RL. Polycystic ovary syndrome as a form of functional ovarian hyperandrogenism due to dysregulation of androgen secretion. Endocr Rev. 1995; 16(3):322-53. https://doi.org/10.1210/er.16.3.322
Franks S, Mason H, Willis D. Follicular dynamics in the polycysticovary syndrome. Mol Cell Endocrinol. 2000; 163(1-2):49-52. https://doi.org/10.1016/S0303- 7207(99)00239-7
Rousseau-Merck M, Atger M, Loosfelt H, Milgrom E, Berger R. Thechromosomal localization of the human Follicle-Stimulating Hormone Receptor gene (FSHR) on 2p21-p16 is similar to that of the luteinizing hormone receptor gene. Genomics. 1993; 15(1):222–4. https://doi. org/10.1006/geno.1993.1041
Dufau ML. The luteinizing hormone receptor. Annu Rev Physiol. 1998; 60(1):461–96. https://doi.org/10.1146/ annurev.physiol.60.1.461
Latronico AC, Lins TSS, Brito VN, Arnhold IJP, Mendonca BB. The effect of distinct activating mutations of the luteinizing hormone receptor gene on the pituitary–gonadal axis in both sexes. Clin Endocrinol. 2000; 53(5):609–13. https://doi.org/10.1046/j.1365-2265.2000.01135.x
Chen Z-J, Zhao H, He L, Shi Y, Qin Y, Shi Y, et al. Genomewide association study identifies susceptibility loci for polycystic ovary syndrome on chromosome 2p16. 3, 2p21 and 9q33. 3. Nat Genet. 2011; 43(1):55–9. https://doi. org/10.1038/ng.732
Mutharasan P, Galdones E, Peñalver Bernabé B, et al. Evidence for chromosome 2p16. 3 polycystic ovary syndrome susceptibility locus in affected women of European ancestry. J Clin Endocrinol Metab. 2013; 98(1):E185–90. https://doi.org/10.1210/jc.2012-2471
Goodarzi MO, Jones MR, Li X, Chua AK, Garcia OA, Chen Y-DI, et al. Replication of association of DENND1A and THADA variants with polycystic ovary syndrome in European cohorts. J Med Genet. 2012; 49(2):90–5. https:// doi.org/10.1136/jmedgenet-2011-100427
Eriksen MB, Brusgaard K, Andersen M, et al. Association of polycystic ovary syndrome susceptibility single nucleotide polymorphism rs2479106 and PCOS in Caucasian patients with PCOS or hirsutism as referral diagnosis. Eur J Obstet Gynecol Reprod Biol. 2012; 163(1):39–42. https://doi. org/10.1016/j.ejogrb.2012.03.020
Welt CK, Styrkarsdottir U, Ehrmann DA, et al. Variants in DENND1A are associated with polycystic ovary syndrome in women of European ancestry. J Clin Endocrinol Metab. 2012; 97(7):E1342–7. https://doi.org/10.1210/jc.2011-3478
Capalbo A, Sagnella F, Apa R, et al. The 312 N variant of the luteinizing hormone/choriogonadotropin receptor gene (LHCGR) confers up to 2· 7-fold increased risk of polycystic ovary syndrome in a Sardinian population. Clin Endocrinol. 2012; 77(1):113–9. https://doi.org/10.1111/ j.1365-2265.2012.04372.x
Singh S, Kaur M, Kaur R, Beri A, Kaur A. Association analysis of LHCGR variants and polycystic ovary syndrome in Punjab: a case-control approach. BMC Endocr Disord. 2022; 22(1):335. https://doi.org/10.1186/s12902-022- 01251-9
Gromoll J, Ried T, Holtgreve-Grez H, Nieschlag E, Gudermann T. Localization of the human FSH receptor to chromosome 2 p21 using a genomic probe comprising exon 10. J Mol Endocrinol. 1994; 12(3):265–71. https://doi. org/10.1677/jme.0.0120265
Gromoll J, Simoni M. Genetic complexity of FSH receptor function. Trends Endocrinol Metab. 2005; 16(8):368–73. https://doi.org/10.1016/j.tem.2005.05.011
Huhtaniemi I. The Parkes lecture. Mutations of gonadotrophin and gonadotrophin receptor genes: what do they teach us about reproductive physiology? Reproduction. 2000; 119(2):173–86. https://doi.org/10.1530/jrf.0.1190173
Saxena R, Georgopoulos NA, Braaten TJ, Bjonnes AC, Koika V, Panidis D, Welt CK. Han Chinese polycystic ovary syndrome risk variants in women of European ancestry: relationship to FSH levels and glucose tolerance. Hum Reprod. 2015; 30(6):1454-9. https://doi.org/10.1093/ humrep/dev085
Dolfin E, Guani B, Lussiana C, et al. FSH receptor Ala307Thr polymorphism is associated to polycystic ovary syndrome and to a higher responsiveness to exogenous FSH in Italian women. J Assist Reprod Genet. 2011; 28(10):925–30. https://doi.org/10.1007/s10815-011-9619-4
Gu B-H, Park J-M, Baek K-H. Genetic variations of follicle stimulating hormone receptor are associated with polycystic ovary syndrome. Int J Mol Med. 2010; 26(1):107–12. https:// doi.org/10.3892/ijmm_00000441
Wu X-Q, Xu S-M, Liu J-F, et al. Association between FSHR polymorphisms and polycystic ovary syndrome among Chinese women in north China. J Assist Reprod Genet. 2014; 31(3):371–7. https://doi.org/10.1007/s10815-013-0166-z
Du J, Zhang W, Guo L, Zhang Z, Shi H, Wang J, et al. Two FSHR variants, haplotypes and meta-analysis in Chinese women with premature ovarian failure and polycystic ovary syndrome. Mol Genet Metab. 2010; 100(3):292–5. https:// doi.org/10.1016/j.ymgme.2010.03.018
Qiu L, Liu J, Hei Q-M. Association between two polymorphisms of follicle stimulating hormone receptor gene and susceptibility to polycystic ovary syndrome: a meta-analysis. Chin Med Sci J. 2015; 30(1):44–50. https:// doi.org/10.1016/S1001-9294(15)30008-0
Janani DM, Ramasubramanyan S, Chellappa V, et al. Whole exome and targeted sequencing reveal novel mutations associated with inherited PCOS condition in an Indian cohort. J Hum Genet. 2023; 68(1):39-46. https://doi. org/10.1038/s10038-022-01093-2
Crespo RP, Rocha TP, Montenegro LR, et al. High-throughput Sequencing to Identify Monogenic Etiologies in a Preselected Polycystic Ovary Syndrome Cohort. J Endocr Soc. 2022; 6(9):bvac106. https://doi.org/10.1210/jendso/bvac106
Naor Z. Signaling by G-protein-coupled receptor (GPCR): studies on the GnRH receptor. Front Neuroendocrinol. 2009; 30(1):10–29. https://doi.org/10.1016/j.yfrne.2008.07.001
Aguilar-Rojas A, Huerta-Reyes M. Human gonadotropinreleasing hormone receptor-activated cellular functions and signaling pathways in extra-pituitary tissues and cancer cells. Oncol Rep. 2009; 22(5):981–90. https://doi. org/10.3892/or_00000525
Ciccone NA, Kaiser UB. The biology of gonadotroph regulation. Curr Opin Endocrinol Diabetes Obes. 2009; 16(4):321. https://doi.org/10.1097/MED.0b013e32832d88fb
Cohen DP, Stein EM, Li Z, Matulis CK, Ehrmann DA, Layman LC. Molecular analysis of the gonadotropinreleasing hormone receptor in patients with polycystic ovary syndrome. Fertil Steril. 1999; 72:360–3. https://doi. org/10.1016/S0015-0282(99)00225-3
Li Q, Yang G, Wang Y, et al. Common genetic variation in the 3′-untranslated region of gonadotropin-releasing hormone receptor regulates gene expression in cella and is associated with thyroid function, insulin secretion as well as insulin sensitivity in polycystic ovary syndrome patients. Hum Genet. 2011; 129:553–61. https://doi.org/10.1007/s00439- 011-0954-4
Caburet S, Fruchter RB, Legois B, Fellous M, Shalev S, Veitia RA. A homozygous mutation of GNRHR in a familial case diagnosed with polycystic ovary syndrome. Eur J Endocrinol. 2017; 176(5):K9-14. https://doi.org/10.1530/ EJE-16-0968
Dumesic DA, Oberfield SE, Stener-Victorin E, et al. Scientific Statement on the Diagnostic Criteria, Epidemiology, Pathophysiology, and Molecular Genetics of Polycystic Ovary Syndrome. Endocr Rev. 2015; 36(5):487- 525. https://doi.org/10.1210/er.2015-1018
Fauser BC, Van Heusden AM. Manipulation of human ovarian function: physiological concepts and clinical consequences. Endocr Rev. 1997; 18:71–106. https://doi. org/10.1210/edrv.18.1.0290
Broekmans FJ, Visser JA, Laven JS, Broer SL, Themmen AP, Fauser BC. Anti-Müllerian hormone and ovarian dysfunction. Trends Endocrinol Metab. 2008; 19(9):340-7. https://doi.org/10.1016/j.tem.2008.08.002
Valkenburg O, Uitterlinden AG, Piersma D, et al. Genetic polymorphisms of GnRH and gonadotrophic hormone receptors affect the phenotype of polycystic ovary syndrome. Hum Reprod. 2009; 24(8):2014-22. https://doi. org/10.1093/humrep/dep113
Josso N, Picard JY, Rey R, di Clemente N. Testicular antiMüllerian hormone: history, genetics, regulation and clinical applications. Pediatr Endocrinol Rev. 2006; 3(4):347-58.
Laven JS, Mulders AG, Visser JA, et al. Anti-Müllerian hormone serum concentrations in normo-ovulatory and anovulatory women of reproductive age. J Clin Endocrinol Metab. 2004; 89(1):318-23. https://doi.org/10.1210/jc.2003- 030932
Stubbs SA, Hardy K, Da Silva-Buttkus P, et al. Anti-müllerian hormone protein expression is reduced during the initial stages of follicle development in human polycystic ovaries. J Clin Endocrinol Metab. 2005; 90(10):5536-43. https://doi. org/10.1210/jc.2005-0907
Visser JA, Durlinger AL, Peters IJ, et al. Increased oocyte degeneration and follicular atresia during the estrous cycle in anti-Müllerian hormone null mice. Endocrinology. 2007; 148(5):2301-8. https://doi.org/10.1210/en.2006-1265
Catteau-Jonard S, Dewailly D. Pathophysiology of polycystic ovary syndrome: the role of hyperandrogenism. Front Horm Res. 2013; 40:22–7. https://doi.org/10.1159/000341679
Cimino I, Casoni F, Liu X, et al. Novel role for antiMüllerian hormone in the regulation of GnRH neuron excitability and hormone secretion. Nat Commun. 2016; 7:10055. https://doi.org/10.1038/ncomms10055
Tata B, Mimouni NEH, Barbotin AL, et al. Elevated prenatal antiMüllerian hormone reprograms the fetus and induces polycystic ovary syndrome in adulthood. Nat Med. 2018; 24(6):834-46. https://doi.org/10.1038/s41591- 018-0035-5
van Rooij IA, Broekmans FJ, te Velde ER, et al. Serum antiMüllerian hormone levels: a novel measure of ovarian reserve. Hum Reprod. 2002; 17(12):3065-71. https://doi. org/10.1093/humrep/17.12.3065
de Vet A, Laven JSE, de Jong FH, Themmen APN, Fauser BCJM. Anti-Müllerian hormone serum levels: a putative marker for ovarian aging. Fert Steril. 2002; 77:357-62. https://doi.org/10.1016/S0015-0282(01)02993-4
Kevenaar ME, Meerasahib MF, Kramer P, et al. Serum antimullerian hormone levels reflect the size of the primordial follicle pool in mice. Endocrinology. 2006; 147(7):3228-34. https://doi.org/10.1210/en.2005-1588
Hansen KR, Hodnett GM, Knowlton N, Craig LB. Correlation of ovarian reserve tests with histologically determined primordial follicle number. Fertil Steril. 2011; 95(1):170-5. https://doi.org/10.1016/j.fertnstert.2010.04.006
Dewailly D, Andersen CY, Balen A, et al. The physiology and clinical utility of anti-Mullerian hormone in women. Hum Reprod Update. 2014; 20(3):370-85. https://doi. org/10.1093/humupd/dmt062
Hawkins Bressler L, Steiner A. Anti-Müllerian hormone as a predictor of reproductive potential. Curr Opin Endocrinol Diabetes Obes. 2018; 25(6):385-90. https://doi.org/10.1097/ MED.0000000000000440
Granger E, Tal R. Anti-Müllerian hormone and its predictive utility in assisted reproductive technologies outcomes. Clin Obstet Gynecol. 2019; 62(2):238-56. https:// doi.org/10.1097/GRF.0000000000000436
Teede H, Misso M, Tassone EC, et al. Anti-Müllerian hormone in PCOS: a review informing international guidelines. Trends Endocrinol Metab. 2019; 30(7):467-78. https://doi.org/10.1016/j.tem.2019.04.006
Sova H, Unkila-Kallio L, Tiitinen A, et al. Hormone profiling, including Anti-Müllerian Hormone (AMH), for the diagnosis of Polycystic Ovary Syndrome (PCOS) and characterization of PCOS phenotypes. Gynecol Endocrinol. 2019; 35(7):595-600. https://doi.org/10.1080/09513590.201 8.1559807
Halder A, Kumar H, Sharma M, Jain M, Kalsi AK. Serum Anti-Müllerian Hormone (AMH): most potential biomarker of PCOS from North India. Ind J Med Res. 2023.
Fallat ME, Siow Y, Marra M, Cook C, Carrillo A. Müllerian inhibiting substance in follicular fluid and serum: a comparison of patients with tubal factor infertility, polycystic ovary syndrome, and endometriosis. Fertil Steril. 1997; 67(5):962-5. https://doi.org/10.1016/S0015- 0282(97)81417-3
Pellatt L, Hanna L, Brincat M, et al. Granulosa cell production of anti-Müllerian hormone is increased in polycystic ovaries. J Clin Endocrinol Metab. 2007; 92(1):240-5. https://doi.org/10.1210/jc.2006-1582
Catteau-Jonard S, Jamin SP, Leclerc A, Gonzalès J, Dewailly D, di Clemente N. Anti-Mullerian hormone, its receptor, FSH receptor, and androgen receptor genes are overexpressed by granulosa cells from stimulated follicles in women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2008; 93(11):4456-61. https:// doi.org/10.1210/jc.2008-1231
Peigné M, Catteau-Jonard S, Robin G, Dumont A, Pigny P, Dewailly D. The numbers of 2-5 and 6-9 mm ovarian follicles are inversely correlated in both normal women and in polycystic ovary syndrome patients: what is the missing link? Hum Reprod. 2018; 33(4):706-14. https:// doi.org/10.1093/humrep/dey017
Wongwananuruk T, Panichyawat N, Indhavivadhana S, et al. Accuracy of anti-Müllerian hormone and total follicles count to diagnose polycystic ovary syndrome in reproductive women. Taiwan J Obstet Gynecol. 2018; 57(4):499-506. https://doi.org/10.1016/j.tjog.2018.06.004
Matsuzaki T, Munkhzaya M, Iwasa T, et al. Relationship between serum anti-Mullerian hormone and clinical parameters in polycystic ovary syndrome. Endocr J. 2017; 64(5):531-41. https://doi.org/10.1507/endocrj.EJ16-0501
Kevenaar ME, Laven JS, Fong SL, et al. A functional antimullerian hormone gene polymorphism is associated with follicle number and androgen levels in polycystic ovary syndrome patients. J Clin Endocrinol Metab. 2008; 93(4):1310-6. https://doi.org/10.1210/jc.2007-2205
Gorsic LK, Dapas M, Legro RS, Hayes MG, Urbanek M. Functional genetic variation in the anti-Müllerian hormone pathway in women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2019; 104(7):2855- 74. https://doi.org/10.1210/jc.2018-02178
Dewailly D, Barbotin AL, Dumont A, Catteau-Jonard S, Robin G. Role of anti-Müllerian hormone in the pathogenesis of polycystic ovary syndrome. Front Endocrinol (Lausanne). 2020; 11:641. https://doi. org/10.3389/fendo.2020.00641
Moolhuijsen LME, Visser JA. AMH in PCOS: Controlling the ovary, placenta, or brain? Curr Opin Endocr Metab Res. 2020; 12:91-7. https://doi.org/10.1016/j. coemr.2020.04.006
di Clemente N, Racine C, Pierre A, Taieb J. AntiMüllerian hormone in female reproduction. Endocr Rev. 2021; 42(6):753-82. https://doi.org/10.1210/endrev/ bnab012
Josso N, Rey RA, Picard JY. Anti-Müllerian hormone: a valuable addition to the toolbox of the pediatric endocrinologist. Int J Endocrinol. 2013; 2013:674105. https://doi.org/10.1155/2013/674105
Josso N, Belville C, di Clemente N, Picard JY. AMH and AMH receptor defects in persistent Müllerian duct syndrome. Hum Reprod Update. 2005; 11(4):351-6. https://doi.org/10.1093/humupd/dmi014
Vasaitis TS, Bruno RD, Njar VC. CYP17 inhibitors for prostate cancer therapy. J Steroid Biochem Mol Biol. 2011; 125(1-2):23-31. https://doi.org/10.1016/j. jsbmb.2010.11.005
Franks, S. The ubiquitous polycystic ovary. J Endocrinol. 1991; 129:317–9. https://doi.org/10.1677/joe.0.1290317
Kumar H, Halder A, Sharma M, Kalsi AK, Jain M. Dihydrotestosterone: a potential biomarker of hyperandrogenaemia in PCOS. J Clin and Diag Res. 2022; 16(2):QC09-14.
McAllister JM, Legro RS, Modi BP, Strauss JF 3rd. Functional genomics of PCOS: from GWAS to molecular mechanisms. Trends Endocrinol Metab. 2015; 26:118–24. https://doi.org/10.1016/j.tem.2014.12.004
Nestler JE, Jakubowicz DJ, de Vargas AF, Brik C, Quintero N, Medina F. Insulin stimulates testosterone biosynthesis by human thecal cells from women with polycystic ovary syndrome by activating its own receptor and using inositolglycan mediators as the signal transduction system. J Clin Endocrinol Metab. 1998; 83(6):2001-5. https://doi.org/10.1210/jc.83.6.2001
Diamanti-Kandarakis E, Dunaif A. Insulin resistance and the polycystic ovary syndrome revisited: an update on mechanisms and implications. Endocr Rev. 2012; 33(6):981-1030. https://doi.org/10.1210/er.2011-1034
Yildiz BO, Azziz R. The adrenal and polycystic ovary syndrome. Rev Endocr Metab Disord. 2007; 8:331–42. https://doi.org/10.1007/s11154-007-9054-0
Rosenfield RL, Ehrmann DA. The Pathogenesis of Polycystic Ovary Syndrome (PCOS): The Hypothesis of PCOS as Functional Ovarian Hyperandrogenism Revisited. Endocr Rev. 2016; 37(5):467-520. https://doi. org/10.1210/er.2015-1104
Lachelin GC, Barnett M, Hopper BR, Brink G, Yen SS. Adrenal function in normal women and women with the polycystic ovary syndrome. J Clin Endocrinol Metab. 1979; 49(6):892-8. https://doi.org/10.1210/jcem-49-6- 892
Jehaimi CT, Araiza VC, Batish SD, Brosnan PG. Polycystic ovaries and adrenal insufficiency in a young pubescent female with lipoid congenital adrenal hyperplasia due to splice mutation of the StAR gene: a case report and review of the literature. J Pediatr Endocrinol Metab. 2010; 23(12):1225–31. https://doi.org/10.1515/jpem.2010.196
Barbar É, LeHoux J-G, Lavigne P. Toward the NMR structure of StAR. Mol Cell Endocrinol. 2009; 300(1– 2):89–93. https://doi.org/10.1016/j.mce.2008.12.007
Miller WL. Steroidogenic acute regulatory protein (StAR), a novel mitochondrial cholesterol transporter. Biochim Biophys Acta. 2007; 1771(6):663–76. https://doi. org/10.1016/j.bbalip.2007.02.012
Jakimiuk AJ, Weitsman SR, Navab A, Magoffin DA. Luteinizing hormone receptor, steroidogenesis acute regulatory protein, and steroidogenic enzyme messenger ribonucleic acids are overexpressed in thecal and granulosa cells from polycystic ovaries. J Clin Endocrinol Metab. 2001; 86(3):1318–23. https://doi.org/10.1210/ jc.86.3.1318
Kahsar-Miller MD, Conway-Myers BA, Boots LR, Azziz R. Steroidogenic acute regulatory protein (StAR) in the ovaries of healthy women and those with polycystic ovary syndrome. Am J Obstet Gynecol. 2001; 185(6):1381–7. https://doi.org/10.1067/mob.2001.118656
Nazouri A-S, Khosravifar M, Akhlaghi A-A, Shiva M, Afsharian P. No relationship between most polymorphisms of steroidogenic acute regulatory (StAR) gene with polycystic ovarian syndrome. Int J Reprod Biomed. 2015; 13(12):771. https://doi.org/10.29252/ ijrm.13.12.771
Jahromi MS, Tehrani FR, Noroozzadeh M, Zarkesh M, Ghasemi A, Zadeh-Vakili A. Elevated expression of steroidogenesis pathway genes; CYP17, GATA6 and StAR in prenatally androgenized rats. Gene. 2016; 593(1):167- 71. https://doi.org/10.1016/j.gene.2016.07.067
Salehi Jahromi M, Hill JW, Ramezani Tehrani F, ZadehVakili A. Hypomethylation of specific CpG sites in the promoter region of steroidogeneic genes (GATA6 and StAR) in prenatally androgenized rats. Life Sci. 2018; 207:105-9. https://doi.org/10.1016/j.lfs.2018.05.052
Khodabandeh S, Hosseini A, Khazali H, Azizi V. Interplay between polycystic ovary syndrome and hypothyroidism on serum testosterone, oxidative stress and StAR gene expression in female rats. Endocrinol Diabetes Metab. 2022; 5(5):e359. https:// doi.org/10.1002/edm2.359
Cousin P, Billotte J, Chaubert P, Shaw P. Physical map of 17p13 and the genes adjacent to p53. Genomics. 2000; 63(1):60–8. https://doi.org/10.1006/geno.1999.6062
Kahn S, Hryb D, Nakhla A, Romas N, Rosner W. Beyond carrier proteins: sex hormone-binding globulin is synthesized in target cells. J Endocrinol. 2002; 175:113– 20. https://doi.org/10.1677/joe.0.1750113
Hammond GL. Plasma steroid-binding proteins: primary gatekeepers of steroid hormone action. J Endocrinol. 2016; 230(1):R13–25. https://doi.org/10.1530/JOE-16- 0070
Pugeat M, Crave JC, Tourniaire J, Forest MG. Clinical utility of sex hormone-binding globulin measurement. Horm Res Paediatr. 1996; 45(3–5):148–55. https://doi. org/10.1159/000184778
Davison SL, Bell R, Donath S, Montalto J, Davis SR. Androgen levels in adult females: changes with age, menopause, and oophorectomy. J Clin Endocrinol Metab. 2005; 90(7):3847–53. https://doi.org/10.1210/ jc.2005-0212
Burger HG, Dudley EC, Cui J, Dennerstein L, Hopper JL. A prospective longitudinal study of serum testosterone, dehydroepiandrosterone sulfate, and sex hormonebinding globulin levels through the menopause transition. J Clin Endocrinol Metab. 2000; 85(8):2832–8. https://doi.org/10.1210/jc.85.8.2832
Pasquali R, Vicennati V, Bertazzo D, et al. Determinants of sex hormone—binding globulin blood concentrations in premenopausal and postmenopausal women with different estrogen status. Metabolism. 1997; 46(1):5–9. https://doi.org/10.1016/S0026-0495(97)90159-1
Rannevik G, Jeppsson S, Johnell OA, Bjerre B, LaurellBorulf Y, Svanberg L. A longitudinal study of the perimenopausal transition: altered profiles of steroid and pituitary hormones, SHBG and bone mineral density. Maturitas. 1995; 21(2):103–13. https://doi. org/10.1016/0378-5122(94)00869-9
Ring HZ, Lessov CN, Reed T, et al. Heritability of plasma sex hormones and hormone binding globulin in adult male twins. J Clin Endocrinol Metab. 2005; 90(6):3653–8. https://doi.org/10.1210/jc.2004-1025
Xita N, Tsatsoulis A. Genetic variants of sex hormonebinding globulin and their biological consequences. Mol Cell Endocrinol. 2010; 316(1):60–5. https://doi. org/10.1016/j.mce.2009.08.025
Hogeveen KN, Cousin P, Pugeat M, Dewailly D, Soudan B, Hammond GL. Human sex hormone–binding globulin variants associated with hyperandrogenism and ovarian dysfunction. J Clin Invest. 2002; 109(7):973–81. https:// doi.org/10.1172/JCI0214060
Morel Y, Bristow J, Gitelman SE, Miller WL. Transcript encoded on the opposite strand of the human steroid 21-hydroxylase/complement component C4 gene locus. Proc Natl Acad Sci. 1989; 86(17):6582–6. https://doi. org/10.1073/pnas.86.17.6582
Mellon SH, Miller WL. Extra adrenal steroid 21-hydroxylation is not mediated by P450c21. J Clin Invest. 1989; 84(5):1497–502. https://doi.org/10.1172/ JCI114325
Rice DA, Kronenberg M, Mouw AR, Aitken L, Franklin A, Schimmer B, et al. Multiple regulatory elements determine adrenocortical expression of steroid 21-hydroxylase. J Biol Chem. 1990; 265(14):8052–8. https://doi.org/10.1016/S0021-9258(19)39037-4
Wijesuriya SD, Zhang G, Dardis A, Miller WL. Transcriptional regulatory elements of the human gene for cytochrome P450c21 (steroid 21-hydroxylase) lie within intron 35 of the linked C4B gene. J Biol Chem. 1999; 274(53):38097–106. https://doi.org/10.1074/ jbc.274.53.38097
Witchel SF, Lee PA, Suda-Hartman M, Hoffman EP. Hyperandrogenism and manifesting heterozygotes for 21-hydroxylase deficiency. Biochem Mol Med. 1997; 62(2):151–8. https://doi.org/10.1006/bmme.1997.2632
Escobar-Morreale HF, San Millán JL, Smith RR, Sancho J, Witchel SF. The presence of the 21-hydroxylase deficiency carrier status in hirsute women: phenotype-genotype correlations. Fertil Steril. 1999; 72(4):629–38. https://doi. org/10.1016/S0015-0282(99)00317-9
Witchel SF, Aston CE. The role of heterozygosity for CYP21 in the polycystic ovary syndrome. J Pediatr Endocrinol Metab. 2000; 13(Suppl 5):1315–7.
Deneux C, Tardy V, Dib A, Mornet E, Billaud L, Charron D, et al. Phenotype-genotype correlation in 56 women with nonclassical congenital adrenal hyperplasia due to 21-hydroxylase deficiency. Journal of Clinical Endocrinology and Metabolism. 2001; 86(1):207–13. https://doi.org/10.1210/jcem.86.1.7131
Chung B-C, Matteson KJ, Voutilainen R, Mohandas T, Miller WL. Human cholesterol side-chain cleavage enzyme, P450scc: cDNA cloning, assignment of the gene to chromosome 15, and expression in the placenta. Proc Natl Acad Sci. 1986; 83(23):8962–6. https://doi. org/10.1073/pnas.83.23.8962
Heidarzadehpilehrood R, Pirhoushiaran M, Abdollahzadeh R, Binti Osman M, Sakinah M, Nordin N, Abdul Hamid H. A Review on CYP11A1, CYP17A1, and CYP19A1 Polymorphism Studies: Candidate Susceptibility Genes for Polycystic Ovary Syndrome (PCOS) and Infertility. Genes (Basel). 2022; 13(2):302. https://doi.org/10.3390/genes13020302
Miller WL. Androgen biosynthesis from cholesterol to DHEA. Mol Cell Endocrinol. 2002; 198(1–2):7–14. https://doi.org/10.1016/S0303-7207(02)00363-5
Shan B, Zhou L, Yang S, Yan M, Wang Z, Ouyang Y, et al. Association between Polycystic Ovary Syndrome (PCOS) and CYP11A1 polymorphism in Hainan, China: a casecontrol study. Int J Clin Exp Pathol. 2016; 9(1):230–6.
Gharani N, Waterworth DM, Batty S, et al. Association of the steroid synthesis gene CYP11a with polycystic ovary syndrome and hyperandrogenism. Hum Mol Genet. 1997; 6(3):397–402. https://doi.org/10.1093/hmg/6.3.397
Shen W, Li T, Hu Y, Liu H, Song M. Common polymorphisms in the CYP1A1 and CYP11A1 genes and polycystic ovary syndrome: a meta-analysis and metaregression. Arch Gynecol Obstet. 2014; 289(1):107–18. https://doi.org/10.1007/s00404-013-2939-0
Diamanti-Kandarakis E, Bartzis MI, Bergiele AT, Tsianateli TC, Kouli CR. Microsatellite polymorphism (tttta) n at- 528 base pairs of gene CYP11α influences hyperandrogenemia in patients with polycystic ovary syndrome. Fertil Steril. 2000; 73(4):735–41. https://doi. org/10.1016/S0015-0282(99)00628-7
Yu M, Feng R, Sun X, Wang H, Wang H, Sang Q, et al. Polymorphisms of pentanucleotide repeats (tttta) n in the promoter of CYP11A1 and their relationships to polycystic ovary syndrome (PCOS) risk: a metaanalysis. Mol Biol Rep. 2014; 41(7):4435–45. https://doi. org/10.1007/s11033-014-3314-3
Rosenfield RL, Barnes RB, Cara JF, Lucky AW. Dysregulation of cytochrome P450c17α as the cause of polycystic ovarian syndrome. Fertil Steril. 1990; 53:785– 91. https://doi.org/10.1016/S0015-0282(16)53510-9
Prapas N, Karkanaki A, Prapas I, Kalogiannidis I, Katsikis I, Panidis D. Genetics of polycystic ovary syndrome. Hippokratia. 2009; 13(4):216.
Sasano H, Okamoto M, Mason J,et al. Immunolocalization of aromatase, 17α-hydroxylase and sidechain-cleavage cytochromes P-450 in the human ovary. Reproduction. 1989; 85(1):163–9. https://doi.org/10.1530/jrf.0.0850163
Tamura T, Kitawaki J, Yamamoto T, et al. Immunohistochemical localization of 17α-hydroxylase/ C17–20 lyase and aromatase cytochrome P-450 in polycystic human ovaries. J Endocrinol. 1993; 139(3):503. https://doi.org/10.1677/joe.0.1390503
Wickenheisser JK, Quinn PG, Nelson VL, et al. Differential activity of the cytochrome P450 17α-hydroxylase and steroidogenic acute regulatory protein gene promoters in normal and polycystic ovary syndrome theca cells. J Clin Endocrinol Metab. 2000; 85(6):2304–11. https://doi. org/10.1210/jcem.85.6.6631
Wickenheisser JK, Nelson-DeGrave VL, Quinn PG, McAllister JM. Increased cytochrome P450 17α-hydroxylase promoter function in theca cells isolated from patients with polycystic ovary syndrome involves nuclear factor-1. Mol Endocrinol. 2004; 18(3):588–605. https://doi.org/10.1210/me.2003-0090
Wickenheisser JK, Nelson-DeGrave VL, McAllister JM. Dysregulation of cytochrome P450 17α-hydroxylase messenger ribonucleic acid stability in theca cells isolated from women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2005; 90(3):1720–7. https://doi. org/10.1210/jc.2004-1860
Yanase T. 17α-hydroxylase/17, 20-lyase defects. J Steroid Biochem Mol Biol. 1995; 53(1–6):153–7. https://doi. org/10.1016/0960-0760(95)00029-Y
Franks S, White D, Gilling-Smith C, et al. Hypersecretion of androgens by polycystic ovaries: the role of genetic factors in the regulation of cytochrome P450c17α. Baillieres Clin Endocrinol Metab. 1996; 10(2):193–203. https://doi.org/10.1016/S0950-351X(96)80057-7
Carey AH, Waterworth D, Patel K, et al. Polycystic ovaries and premature male pattern baldness are associated with one allele of the steroid metabolism gene CYP17. Hum Mol Genet. 1994; 3(10):1873–6. https://doi.org/10.1093/ hmg/3.10.1873
Miyoshi Y, Iwao K, Ikeda N, Egawa C, Noguchi S. Genetic polymorphism in CYP17 and breast cancer risk in Japanese women. Eur J Cancer. 2000; 36(18):2375–9. https://doi.org/10.1016/S0959-8049(00)00334-8
Li Y, Liu F, Luo S, Hu H, Li X-H, Li S-W. Polymorphism T→ C of gene CYP17 promoter and polycystic ovary syndrome risk: a meta-analysis. Gene. 2012; 495(1):16– 22. https://doi.org/10.1016/j.gene.2011.12.048
Pusalkar M, Meherji P, Gokral J, Chinnaraj S, Maitra A. CYP11A1 and CYP17 promoter polymorphisms associate with hyperandrogenemia in polycystic ovary syndrome. Fertil Steril. 2009; 92(2):653–9. https://doi. org/10.1016/j.fertnstert.2008.07.016
Chen S, Besman MJ, Sparkes RS, et al. Human aromatase: cDNA cloning, southern blot analysis, and assignment of the gene to chromosome 15. DNA. 1988; 7(1):27–38. https://doi.org/10.1089/dna.1988.7.27
Takayama K, Suzuki T, Bulun SE, Sasano H, Yilmaz B, Sebastian S, editors. Organization of the human aromatase p450 (CYP19) gene. In: Seminars in reproductive medicine. New York: Copyright© 2004 by Thieme Medical Publishers, Inc; 2004.
Simpson ER, Mahendroo MS, Means GD, et al. Aromatase cytochrome P450, the enzyme responsible for estrogen biosynthesis. Endocr Rev. 1994; 15(3):342–55. https:// doi.org/10.1210/er.15.3.342
Graham-Lorence S, Khalil MW, Lorence MC, Mendelson CR, Simpson ER. Structure-function relationships of human aromatase cytochrome P-450 using molecular modeling and site-directed mutagenesis. J Biol Chem. 1991; 266(18):11939–46. https://doi.org/10.1016/S0021- 9258(18)99048-4
Hickey GJ, Chen S, Besman MJ, et al. Hormonal regulation, tissue distribution, and content of aromatase cytochrome P450 messenger ribonucleic acid and enzyme in rat ovarian follicles and corpora lutea: relationship to estradiol biosynthesis. Endocrinology. 1988; 122(4):1426–36. https://doi.org/10.1210/endo- 122-4-1426
de Medeiros SF, Barbosa JS, Yamamoto MMW. Comparison of steroidogenic pathways among normoandrogenic and hyperandrogenic polycystic ovary syndrome patients and normal cycling women. J Obstet Gynaecol Res. 2015;41(2):254–63. https://doi. org/10.1111/jog.12524
Petry C, Ong K, Michelmore K, Artigas S, Wingate D, Balen A, et al. Association of aromatase (CYP 19) gene variation with features of hyperandrogenism in two populations of young women. Hum Reprod. 2005; 20(7):1837–43. https://doi.org/10.1093/humrep/deh900
Yu Y-Y, Sun C-X, Liu Y-K, Li Y, Wang L, Zhang W, et al. Promoter methylation of CYP19A1 gene in Chinese polycystic ovary syndrome patients. Gynecol Obstet Investig. 2013; 76(4):209–13. https://doi. org/10.1159/000355314
Sowers MR, Wilson AL, Kardia SR, Chu J, Ferrell R. Aromatase gene (CYP 19) polymorphisms and endogenous androgen concentrations in a multiracial/ multiethnic, multisite study of women at midlife. Am J Med. 2006; 119(9):S23–30. https://doi.org/10.1016/j. amjmed.2006.07.003
Ranjith R, Rani U, Nagarajeshwari C, Unnisa W, Nalini S, Jahan P. Genetics: androgen associated gene polymorphism (s) in women with polycystic ovary syndrome from South Indian population; 2011.
Al-Salihi AR, Hamdan FB, Mutib MT. Effect of CYP19 gene on polycystic ovary syndrome phenotype in Iraqi women. Iraqi J Med Sci. 2015; 13(3):272–8.
Mehdizadeh A, Kalantar SM, Sheikhha MH, Aali BS, Ghanei A. Association of SNP rs. 2414096 CYP19 gene with polycystic ovarian syndrome in Iranian women. Int J Reprod Biomed. 2017; 15(8):491. https://doi. org/10.29252/ijrm.15.8.491
Xu P, Zhang X, Xie G, et al. The (TTTA) n polymorphism in intron 4 of CYP19 and the polycystic ovary syndrome risk in a Chinese population. Mol Biol Rep. 2013; 40(8):5041–7. https://doi.org/10.1007/s11033-013- 2605-4
Mostafa RA, Al-Sherbeeny MM, Abdelazim IA, et al. Relation between aromatase gene CYP19 variation and hyperandrogenism in polycystic ovary syndrome Egyptian women. J Infertil Reprod Biol. 2016; 4:1–5.
Lazaros L, Xita N, Hatzi E, et al. CYP19 gene variants affect the assisted reproduction outcome of women with polycystic ovary syndrome. Gynecol Endocrinol. 2013; 29(5):478–82. https://doi.org/10.3109/09513590.2013.77 4359
Xita N, Lazaros L, Georgiou I, Tsatsoulis A. CYP19 gene: a genetic modifier of polycystic ovary syndrome phenotype. Fertil Steril. 2010; 94(1):250–4. https://doi. org/10.1016/j.fertnstert.2009.01.147
Chamberlain NL, Driver ED, Miesfeld RL. The length and location of CAG trinucleotide repeats in the androgen receptor N-terminal domain affect transactivation function. Nucleic Acids Res. 1994; 22(15):3181–6. https:// doi.org/10.1093/nar/22.15.3181
Adams J, Polson D, Franks S. Prevalence of polycystic ovaries in women with anovulation and idiopathic hirsutism. Br Med J (Clin Res Ed). 1986; 293(6543):355– 9. https://doi.org/10.1136/bmj.293.6543.355
Xita N, Tsatsoulis A. Fetal programming of polycystic ovary syndrome by androgen excess: evidence from experimental, clinical, and genetic association studies. J Clin Endocrinol Metab. 2006; 91(5):1660–6. https://doi. org/10.1210/jc.2005-2757
Zeng X, Zhong Q, Li M, Liu Y, Long S, Xie Y, Mo Z. Androgen increases klotho expression via the androgen receptor-mediated pathway to induce GCs apoptosis. J Ovarian Res. 2023; 16(1):10. https://doi.org/10.1186/ s13048-022-01087-w
Walters K, Allan C, Handelsman D. Androgen actions and the ovary. Biol Reprod. 2008; 78(3):380–9. https:// doi.org/10.1095/biolreprod.107.064089
Schüring A, Welp A, Gromoll J, Zitzmann M, Sonntag B, Nieschlag E, et al. Role of the CAG repeat polymorphism of the androgen receptor gene in Polycystic Ovary Syndrome (PCOS). Exp Clin Endocrinol Diabetes. 2012; 120(2):73s. https://doi.org/10.1055/s-0031-1291343
Xia Y, Che Y, Zhang X, Zhang C, Cao Y, Wang W, et al. Polymorphic CAG repeat in the androgen receptor gene in polycystic ovary syndrome patients. Mol Med Rep. 2012; 5(5):1330–4.
Lin LH, Baracat MC, Maciel GA, Soares JM Jr, Baracat EC. Androgen receptor gene polymorphism and polycystic ovary syndrome. Int J Gynecol Obstet. 2013; 120(2):115–8. https://doi.org/10.1016/j.ijgo.2012.08.016
Shah NA, Antoine HJ, Pall M, et al. Association of androgen receptor CAG repeat polymorphism and polycystic ovary syndrome. J Clin Endocrinol Metab. 2008; 93(5):1939–45. https://doi.org/10.1210/jc.2008-0038
Mifsud A, Ramirez S, Yong E. Androgen receptor gene CAG trinucleotide repeats in anovulatory infertility and polycystic ovaries. J Clin Endocrinol Metab. 2000; 85(9):3484–8. https://doi.org/10.1210/jcem.85.9.6832
Rajender S, Carlus SJ, Bansal SK, et al. Androgen receptor CAG repeats length polymorphism and the risk of Polycystic Ovarian Syndrome (PCOS). PLoS One. 2013; 8(10):e75709. https://doi.org/10.1371/journal. pone.0075709
Ferk P, Perme MP, Teran N, Gersak K. Androgen receptor gene (CAG) n polymorphism in patients with polycystic ovary syndrome. Fertil Steril. 2008; 90(3):860–3. https:// doi.org/10.1016/j.fertnstert.2007.07.1291
Kim JJ, Choung SH, Choi YM, Yoon SH, Kim SH, Moon SY, et al. Androgen receptor gene CAG repeat polymorphism in women with polycystic ovary syndrome. Fertil Steril. 2008; 90(6):2318–23. https://doi. org/10.1016/j.fertnstert.2007.10.030
Skrgatic L, Baldani DP, Cerne J, Ferk P, Gersak K. CAG repeat polymorphism in androgen receptor gene is not directly associated with polycystic ovary syndrome but influences serum testosterone levels. J Steroid Biochem Mol Biol. 2012; 128 (3–5):107–12. https://doi. org/10.1016/j.jsbmb.2011.11.006
Lim JJ, Lima PDA, Salehi R, Dong RL, Tsang BK. Regulation of androgen receptor signaling by ubiquitination during folliculogenesis and its possible dysregulation in polycystic ovarian syndrome. Sci Rep. 2017; 7(1):10272. https://doi.org/10.1038/s41598-017- 09880-0
Walters KA, Middleton LJ, Joseph SR, et al. Targeted loss of androgen receptor signaling in murine granulosa cells of preantral and antral follicles causes female subfertility. Biol Reprod. 2012; 87(6):151. https://doi.org/10.1095/ biolreprod.112.102012
Walters KA, Edwards MC, Tesic D, et al. The role of central androgen receptor actions in regulating the hypothalamicpituitary-ovarian axis. Neuroendocrinology. 2018; 106(4):389–400. https://doi.org/10.1159/000487762
Tian L, Zou Y, Tan J, et al. Androgen receptor gene mutations in 258 Han Chinese patients with polycystic ovary syndrome. Exp Ther Med. 2021; 21(1):31. https:// doi.org/10.3892/etm.2020.9463
Kyrönlahti A, Vetter M, Euler R, et al. GATA4 deficiency impairs ovarian function in adult mice. Biol Reprod. 2011; 84:1033-44. https://doi.org/10.1095/ biolreprod.110.086850
Padua MB, Fox SC, Jiang T, Morse DA, Tevosian SG. Simultaneous gene deletion of gata4 and gata6 leads to early disruption of follicular development and germ cell loss in the murine ovary. Biol Reprod. 2014; 91(24):1-10. https://doi.org/10.1095/biolreprod.113.117002
LaVoie HA, Singh D, Hui YY. Concerted regulation of the porcine steroidogenic acute regulatory protein gene promoter activity by follicle-stimulating hormone and insulin-like growth factor I in granulosa cells involves GATA-4 and CCAAT/enhancer binding protein β. Endocrinology. 2004; 145:3122-34. https://doi. org/10.1210/en.2003-1719
Kwintkiewicz J, Cai Z, Stocco C. Follicle-stimulating hormone-induced activation of Gata4 contributes in the up-regulation of Cyp19 expression in rat granulosa cells. Mol Endocrinol. 2007; 21:933-47. https://doi. org/10.1210/me.2006-0446
Sharma P, Jain M, Halder A. An Investigation of Steroid Biosynthesis Pathway Genes in Women with Polycystic Ovary Syndrome. J Hum Reprod Sci. 2022; 15(3):240-9. https://doi.org/10.4103/jhrs.jhrs_86_22
Bergman RN, Ider YZ, Bowden CR, Cobelli C. Quantitative estimation of insulin sensitivity. Am J Physiol. 1979; 236:E667. https://doi.org/10.1152/ ajpendo.1979.236.6.E667
O’Meara NM, Blackman JD, Ehrmann DA, Barnes RB, Jaspan JB, Rosenfield RL, Polonsky KS. Defects in betacell function in functional ovarian hyperandrogenism. J Clin Endocrinol Metab. 1993; 76(5):1241-7. https://doi. org/10.1210/jcem.76.5.8496316
Ehrmann DA, Sturis J, Byrne MM, Karrison T, Rosenfield RL, Polonsky KS. Insulin secretory defects in polycystic ovary syndrome. Relationship to insulin sensitivity and family history of non-insulin-dependent diabetes mellitus. J Clin Invest. 1995; 96(1):520-7. https://doi. org/10.1172/JCI118064
Dunaif A, Finegood DT. Beta-cell dysfunction independent of obesity and glucose intolerance in the polycystic ovary syndrome. J Clin Endocrinol Metab. 1996; 81(3):942-7. https://doi.org/10.1210/jcem.81.3.8772555 214. Pasquali R, Venturoli S, Paradisi R, Capelli M, Parenti M, Melchionda N. Insulin and C-peptide levels in obese patients with polycystic ovaries. Horm Metab Res. 1982; 14(6):284-7. https://doi. org/10.1055/s-2007-1018996
Mahabeer S, Jialal I, Norman RJ, Naidoo C, Reddi K, Joubert SM. Insulin and C-peptide secretion in non-obese patients with polycystic ovarian disease. Horm Metab Res. 1989; 21(9):502-6. https://doi. org/10.1055/s-2007-1009272
Ciaraldi TP, el-Roeiy A, Madar Z, Reichart D, Olefsky JM, Yen SS. Cellular mechanisms of insulin resistance in polycystic ovarian syndrome. J Clin Endocrinol Metab. 1992; 75(2):577-83. https://doi.org/10.1210/ jcem.75.2.1322430
Corbould A, Kim YB, Youngren JF, Pender C, Kahn BB, Lee A, Dunaif A. Insulin resistance in the skeletal muscle of women with PCOS involves intrinsic and acquired defects in insulin signaling. Am J Physiol Endocrinol Metab. 2005; 288(5):E1047-54. https://doi.org/10.1152/ ajpendo.00361.2004
Ciaraldi TP, Aroda V, Mudaliar S, Chang RJ, Henry RR. Polycystic ovary syndrome is associated with tissue-specific differences in insulin resistance. J. Clin. Endocrinol. Metab. 2009; 94:157–63. https://doi. org/10.1210/jc.2008-1492
El Mkadem SA, Lautier C, Macari F, et al. Role of allelic variants gly972arg of irs-1 and gly1057asp of irs-2 in moderate-to-severe insulin resistance of women with polycystic ovary syndrome. Diabetes. 2001; 50(9):2164-8. https://doi.org/10.2337/diabetes.50.9.2164
Diamanti-Kandarakis E., Papavassiliou A.G. Molecular mechanisms of insulin resistance in polycystic ovary syndrome. Trends Mol Med. 2006; 12:324–32. https://doi. org/10.1016/j.molmed.2006.05.006
Shi X, Xie X, Jia Y, Li S. Associations of insulin receptor and insulin receptor substrates genetic polymorphisms with polycystic ovary syndrome: A systematic review and meta-analysis. J Obstet Gynaecol Res. 2016; 42(7):844- 54. https://doi.org/10.1111/jog.13002
Feigelson HS, Teras LR, Diver WR, Tang W, et al. Genetic variation in candidate obesity genes ADRB2, ADRB3, GHRL, HSD11B1, IRS1, IRS2, and SHC1 and risk for breast cancer in the Cancer Prevention Study II. Breast Cancer Res. 2008; 10:R57. https://doi.org/10.1186/ bcr2114
Ruan Y, Ma J, Xie X. Association of IRS-1 and IRS-2 genes polymorphisms with polycystic ovary syndrome: a meta-analysis. Endocr J. 2012; 59(7):601-9. https://doi. org/10.1507/endocrj.EJ11-0387
Perez-Martinez P, Delgado-Lista J, Garcia-Rios A, et al. Insulin receptor substrate-2 gene variants in subjects with metabolic syndrome: Association with plasma monounsaturated and n-3 polyunsaturated fatty acid levels and insulin resistance. Mol Nutr Food Res. 2012; 56:309-15. https://doi.org/10.1002/ mnfr.201100504
Gu H, Li L, Zhou B, Li M, Zhong W, Wei X, Zhong X. Single nucleotide polymorphisms in binding site of miRNA-135a and targeted gene IRS2 are correlated with multiple clinical features of PCOS: A study in Chinese women. Technol Health Care. 2022; 30(S1):71-80. https:// doi.org/10.3233/THC-228007
Seino S, Seino M, Bell GI. Human insulin-receptor gene. Diabetes. 1990; 39(2):129–33. https://doi.org/10.2337/ diab.39.2.129
Accili D, Drago J, Lee EJ, et al. Early neonatal death in mice homozygous for a null allele of the insulin receptor gene. Nat. Genet. 1996; 12:106–9. https://doi.org/10.1038/ ng0196-106
Rager KM, Omar HA. Androgen excess disorders in women: the severe insulin-resistant hyperandrogenic syndrome, HAIR-AN. Sci World J. 2006; 6:116–21. https://doi.org/10.1100/tsw.2006.23
Feng C, Lv PP, Yu TT, Jin M, Shen JM, Wang X, Zhou F, Jiang SW. The association between polymorphism of INSR and polycystic ovary syndrome: a meta-analysis. Int J Mol Sci. 2015; 16(2):2403-25. https://doi.org/10.3390/ijms16022403
Siegel S, Futterweit W, Davies TF, Concepcion ES, Greenberg DA, Villanueva R, Tomer Y. A C/T single nucleotide polymorphism at the tyrosine kinase domain of the insulin receptor gene is associated with polycystic ovary syndrome. Fertil Steril. 2002; 78:1240–3. https:// doi.org/10.1016/S0015-0282(02)04241-3
Unsal T, Konac E, Yesilkaya E, et al. Genetic polymorphisms of FSHR, CYP17, CYP1A1, CAPN10, INSR, SERPINE1 genes in adolescent girls with polycystic ovary syndrome. J Assist Reprod Genet. 2009; 26:205–16. https://doi.org/10.1007/s10815-009-9308-8
Ioannidis A, Ikonomi E, Dimou NL, et al. Polymorphisms of the insulin receptor and the insulin receptor substrates genes in polycystic ovary syndrome: A Mendelian randomization meta-analysis. Mol Genet Metable. 2010; 99:174–83. https://doi.org/10.1016/j. ymgme.2009.10.013
Shi Y, Zhao H, Shi Y, et al. Genome-wide association study identifies eight new risk loci for polycystic ovary syndrome. Nat Genet. 2012; 44:1020–5. https://doi. org/10.1038/ng.2384
Feng C, Lv P-P, Yu T-T, Jin M, Shen J-M, Wang X, et al. The association between polymorphism of INSR and polycystic ovary syndrome: a meta-analysis. Int J Mol Sci. 2015; 16(2):2403–25. https://doi.org/10.3390/ ijms16022403
Liu F, Tian L, Tan J, et al. Identification of a novel ESR1 mutation in a Chinese PCOS woman with estrogen insensitivity in IVF treatment. Reprod Biol Endocrinol. 2022; 20(1):157. https://doi.org/10.1186/s12958-022- 01029-7
Tamaoka S, Saito K, Yoshida T, Nakabayashi K, Tatsumi K, et al. Exome-based genome-wide screening of rare variants associated with the risk of polycystic ovary syndrome. Reprod Med Biol. 2023; 22(1):e12504. https:// doi.org/10.1002/rmb2.12504
Karakaya C, Çil AP, Bilguvar K, et al. Further delineation of familial Polycystic Ovary Syndrome (PCOS) via whole-exome sequencing: PCOS-related rare FBN3 and FN1 gene variants are identified. J Obstet Gynaecol Res. 2022; 48(5):1202-11. https://doi.org/10.1111/jog.15187
 Priyal Sharma
Priyal Sharma